Clinical reports come in all shapes, sizes and flavors. With that in mind, our clinical reporting interface VSReports was built to be highly customizable and flexible. With a little Javascript and HTML know-how, your clinical reports can be customized to meet the needs and goals of your lab. With a little Javascript and HTML know how, you can customize yours as well!
Coming up in our next VarSeq release, we’ve updated our report templates to showcase some variability in look, feel and information presented. (Remember, if the information is in the VarSeq table view, it can be added to your report!) We’ve also added a brand new template for Exome Trios which can be easily modified for Single Exomes, or even gene panels. These reports come with your VSReports license, so we encourage you to use them and let us know what you think!
Clinical Reports – Updates and a New Addition
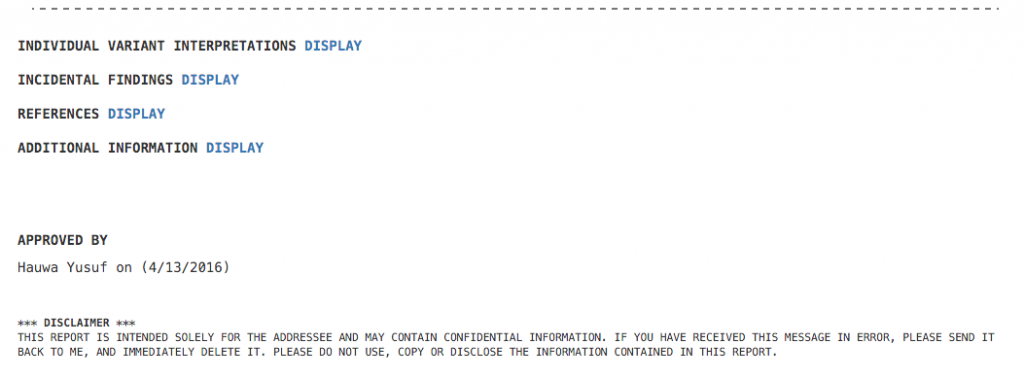
First up, we’ve revamped the ACMG Hereditary Gene Panel Report Template. It now features a clean ASCII-style format, collapsing sections and a disclaimer section. Given BAM files for the sample, this report template automatically calculates average read depth and panel coverage. The ability to expand and collapse sections in this template prevents information overload and presents a minimalistic report view – You can show and hide information as needed.
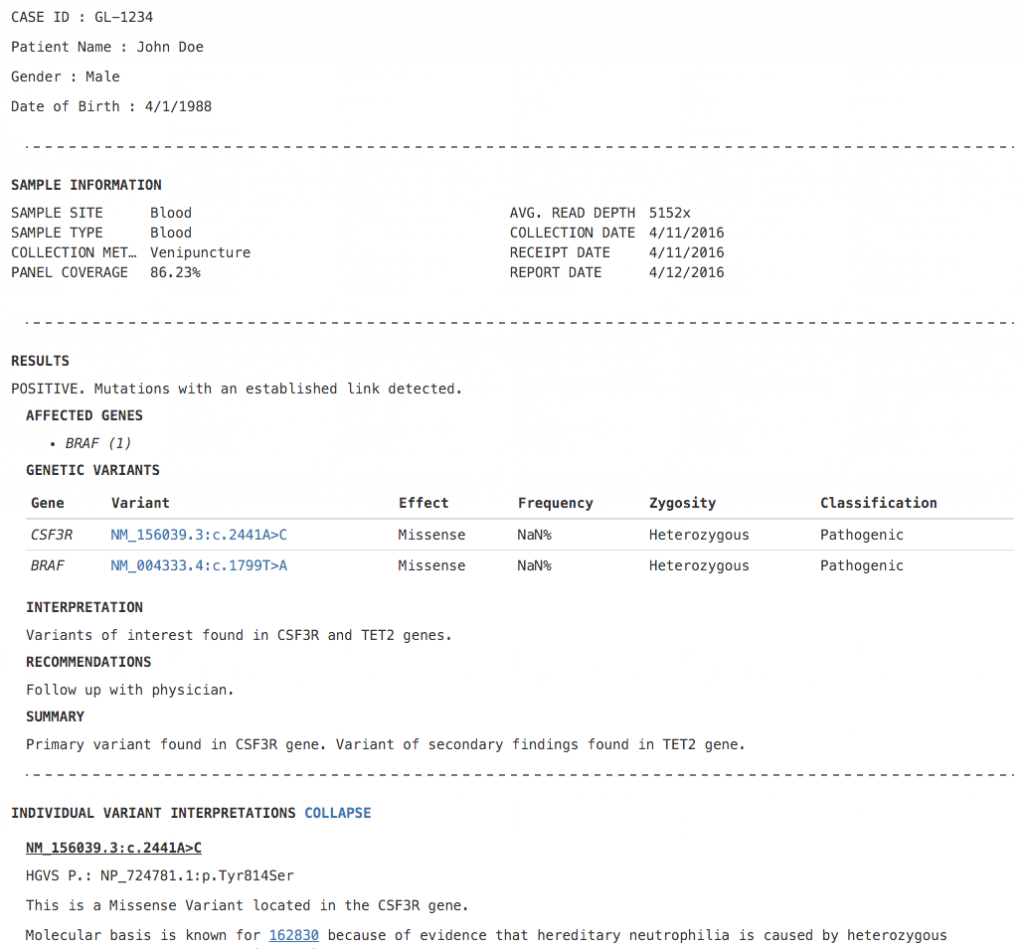
Figure 1: ACMG Hereditary Gene Panel Report Template
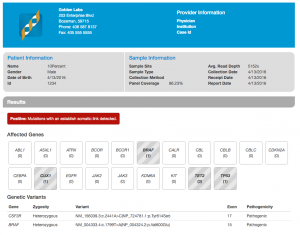
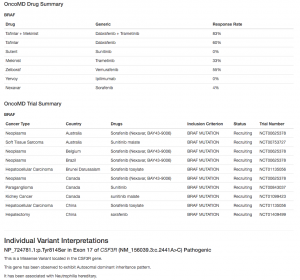
Next, we have our Cancer Gene Panel Report Template. Similar in content to the Hereditary Gene Panel, the Cancer Gene Panel Report Template collects information about our sample, automatically calculating read depth and panel coverage (given sample BAM files), and displays tabular information about our variants of interest. Our Cancer Gene Panel Template also includes Drug information and Trial Summary from OncoMD. We’ve updated the look of the header section to feature white text on a colored background and added a section for your organization’s logo, giving your report a pop of color.
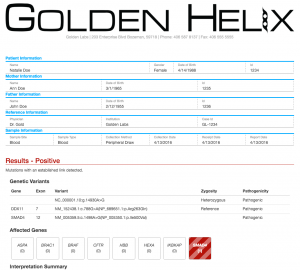
Last but not least, we’ve added a new Exome Trio Report Template. The exome trio report template features a clean and colorful styling that makes skimming sections of the report easier. The header supports a wide width logo for your organization, and patient, mother, father and sample information are displayed in a form-like view.
The results section is interactive as it changes from red to green and vice versa as you change your results from positive to negative. Affected genes are shaded in red.
The bottom of the report showcases a new sign off section that contains an electronic signature and sign off date as well your organization’s CLIA ID.
This report can be easily modified for single exome projects or even gene panels.
We welcome you to use these updated and new reports to your organization’s content. Remember with a some Javascript/HTML know-how, your report can be as customized as you want or need. If you don’t have the resources to help you customize your reports, we’re more than happy to help you!
Contact us at support@goldenhelix.com for more information about customizing your reports.